|
Ion Exchanger |
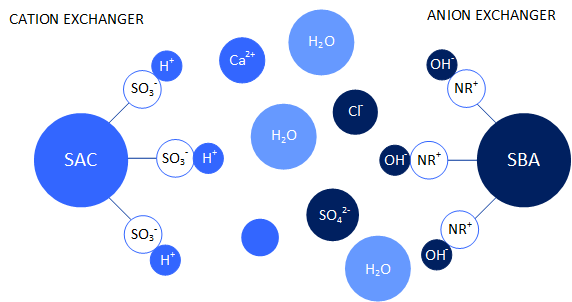
Ion exchange processes are based on the exchange and attachment of ions to synthetic resins, which are made of organic polymers. These synthetic resins contain functional groups to which cations or anions can be attached, depending on the type of ion exchanger. Thus, a basic distinction is made between anion and cation exchangers, which in turn are divided into weak and strong basic or weak and strong acid. The deposition of the ions takes place with a selectivity. For example, Fe3+ is preferred to Mg2+ in the cation exchanger, while Mg2+ is preferred to Na+ and H+.
In regenerated form, a strongly acidic cation exchanger is present in the H+ form. Thus all functional groups are occupied with H+ - ions. If water is now passed over the ion exchanger with a load of ions, ions will be deposited on the cation exchanger, which are located further up in the selectivity series. The H+ - ion is released. The same applies to a strongly alkaline anion exchanger which is present in the regenerated form in the OH- - form. Thus, when strongly acidic cation exchange resins and strongly basic anion exchange resins are mixed, an almost complete separation of ions contained in the water can be achieved.
|
|
If the functional groups in the resin are occupied by the ions to be separated, a salt breakthrough of the resin bed occurs. The resin is thus exhausted and must be regenerated. During regeneration, a solution containing the ions that the resin should have in the regenerated form (e.g. H+) is run over the ion exchange resins in excess. Some of the ions in excess, despite their poorer selectivity, accumulate on the ion exchange resins and displace the ions with a higher selectivity. A high surplus of e.g. H+ ions thus makes it possible to completely convert the ion exchange resin back into the regenerated form.
Our typical applications for ion exchange processes are
- Softening of e.g. drinking water, process water
- Removal of ammonia from condensate systems using cation exchangers
- Complete desalination by means of mixed bed ion exchanger or EDI according to VGB-S-010-T-00 (formerly R 450 L)
- Removal of residual colorants and salts from process solutions
Separate the best from the rest - Individual process combinations for highest efficiency
Ion exchange processes are often used as polishing stages in an overall process. Ion exchangers can effectively remove residual ion loads where membrane technology reaches its limits. Thus, ion exchange technology is an optimal complement to our membrane technology.
The right support in every stage of the project
We have gained experience with a variety of different process fluids. We would be pleased to assist you as a plant engineering company in the selection of the suitable membrane and the optimal process control! For special applications, we are also happy to offer you the possibility of process screening in our laboratory with subsequent industrial piloting as part of our 4 steps 2 separation!